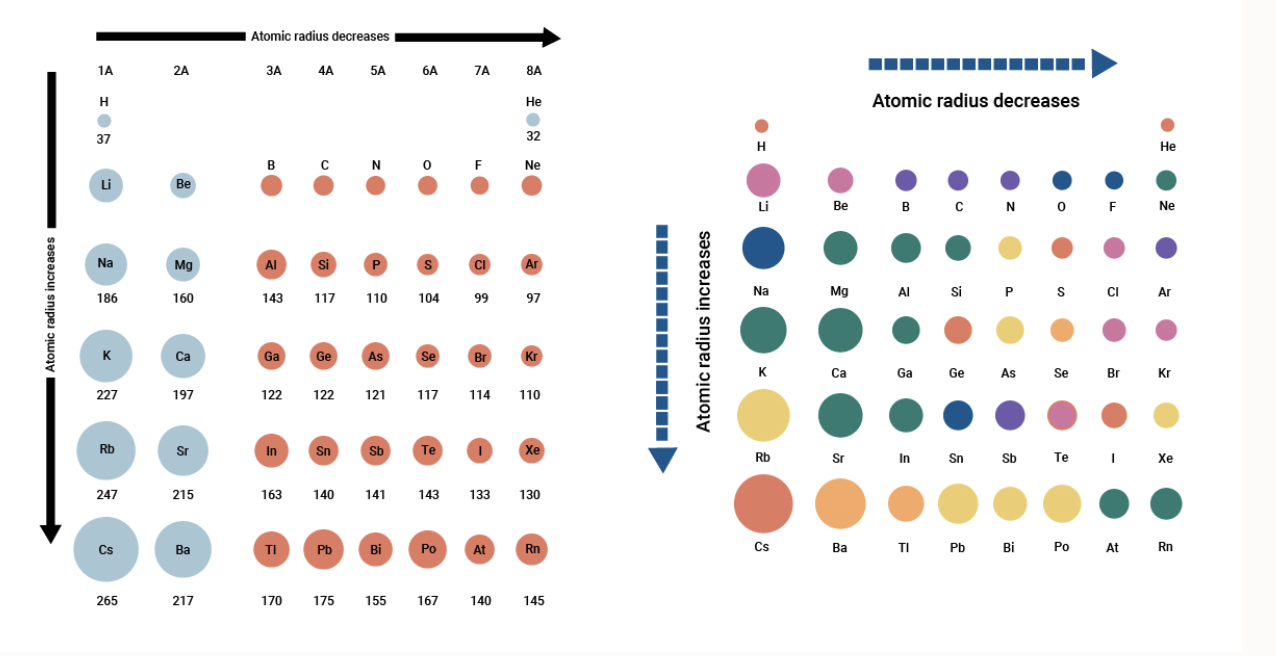
Atomic radius periodic trend9/19/2023  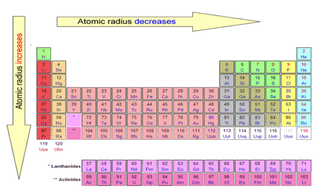
This leads to larger atomic radius going down the group. To reduce this, the outer shell is pushed further away from the nucleus due to the repulsion and so it is less attracted to the nucleus (see #3 above). Being too close to the inner electron shells would cause repulsion (see #4).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
